 
Here, again, Umar is using the full shells explanatory principle as the basis for explaining the bond in terms of electrons 'filling up the gaps' in the electron shells, rather than considering how electrical interactions can hold the structure together. They like, one that fills up the gap, fills up the – last electron needed in one of the chlorine shells, and the other chlorine shell fills it up in the other one. So where do those two electrons come from? So how many electrons are involved in a covalent bond? So how many electrons does each chlorine have to start with? (The outer shell of chlorine would only actually be 'full' with 18 electrons, but that complication is seldom recognised, as octets and full shells are usually considered synonymous by students). In thinking about covalent bonding, Umar (in common with many students) drew upon the full shells explanatory principle that considered bonding to be driven by the needs of atoms to 'fill' their outer electron shells. Like the one on it, the one of the chlorines shares electrons with the other chlorine to fill in its shell on the other one, and the same does it with the other. So how many electrons would they have then? So what would that be, covalently bonded? Now Umar had recalled that chlorine comprises of covalent molecules, I asked him about the nature of the bond: Other slightly different questions may well have cued the elicitation of different knowledge. This suggests that what students respond in written tests should be considered only as what they were triggered to write on that day in response to those particular questions, and may not fully reflect their knowledge and understanding of science topics. Although he suggested the bond was (actually " might be") covalent, this seemed less something that he confidently recalled, than something he was inferring from what he could remember – or perhaps even guessing at what seemed reasonable: " they might be just, they might be like, be covalently bonded".Īs often happens in talking to learners in depth about their ideas it becomes clear that thinking of students 'knowing' or 'not knowing' particular things is a fairly inadequate way of conceptualising their cognition, which is often nuanced and context-dependent. It seemed that my explicit reference to the formula, Cl 2, (eventually) activated his knowledge of the molecule bringing to mind something he had forgotten. Perhaps the earlier context of talking about polar bonds and the trichloroethane molecule somehow acted as a kind of impediment to Umar remembering about the chlorine molecule. They might be just, they might be like, be covalently bonded. So you couldn’t have sort of Cl 2, a molecule of Cl 2? Umar's response here rather surprised me, as I was pretty confident that Umar had met chlorine as an element, and would know it was comprised of diatomic molecules: Cl 2. What about if you just had two chlorine atoms joined together and nothing else, is that possible? You get, it still, you get, if you had like two chlorines it depends what groups are attached to it, to see how electronegative or electropositive they are. What substance would you get if two chlorine atoms formed a bond? Is it possible for chlorine to form a bond with another chlorine? Given his apparently confused notion about the C-Cl bond I decided to turn the conversation to a covalent bond which I knew, well certainly believed, was more familiar to him. Umar then thought this compound would have polar bonding, however he seemed to have difficulty explaining what this meant ⚗︎. When I spoke to him near the start of his second term, I asked him again about this. 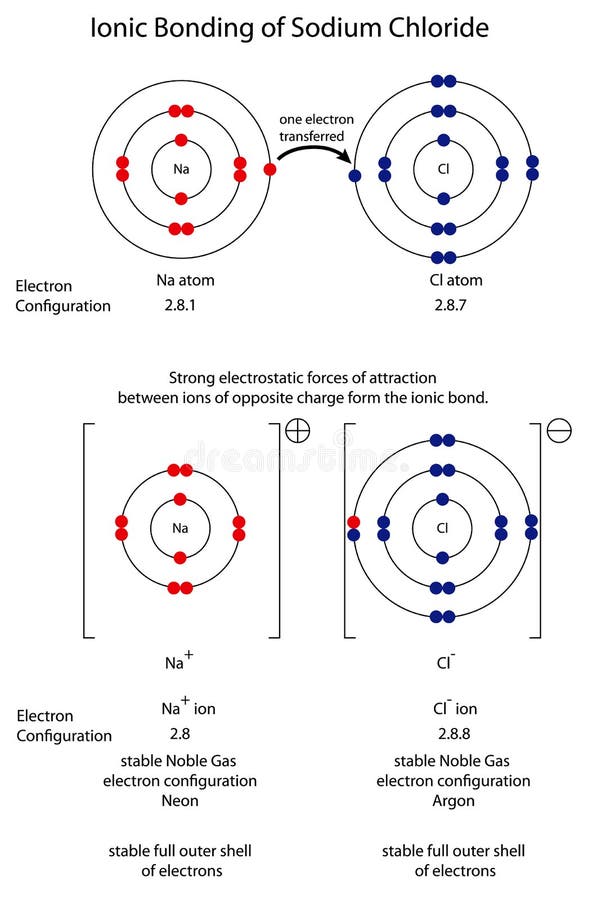
When I spoke to him in the first term of his course he was unsure whether tetrachloromethane (CCl 4) would have ionic or covalent bonding. Umar was a participant in the Understanding Chemical Bonding project.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
